X-ray Crystallography of Oxygen-Tolerant Membrane-Bound [NiFe] Hydrogenase (Press Release)
- Release Date
- 17 Oct, 2011
- BL41XU (Structural Biology I)
- BL44XU (Macromolecular Assemblies)
University of Hyogo
Ibaraki University
RIKEN
|
Yoshiki Higuchi (Professor) and Yasuhito Shomura (Assistant Professor) of the Graduate School of Life Science, University of Hyogo, in cooperation with a research group led by Hirofumi Nishihara (Associate Professor) of the College of Agriculture, Ibaraki University and a group from RIKEN, carried out X-ray crystallography to analyze the steric structure of oxygen-tolerant membrane-bound [NiFe] hydrogenase,*1 an enzyme protein, using SPring-8 beamlines. This is the world’s first analysis of the steric structure of oxygen-tolerant hydrogenase.*2 The standard [NiFe] hydrogenase that has been studied thus far is known to lose its enzymatic function when exposed to oxygen. The structural analysis revealed that part of the oxygen-tolerant membrane-bound [NiFe] hydrogenase molecules induces a characteristic structural change when exposed to oxygen. This structural change explains why the function of the hydrogenase as an enzyme molecule is not lost when the enzyme is exposed to oxygen. The results provide an important knowledge for overcoming the functional loss of hydrogenase upon exposure to oxygen. The results are also expected to be applied to the research and development of applications of hydrogen energy, such as in new chemically synthesized catalysts that are nonreactive to oxygen and in new fuel cells. The results were published online in Nature, a prestigious scientific journal, on 16 October 2011. Publication: |
<<Glossary>>
*1 [NiFe] hydrogenase
[NiFe] hydrogenase is an enzyme contained in many microorganisms and controls the reduction and oxidation of hydrogen molecules. Ni and Fe atoms play an important functional role in this enzyme.
*2 Oxygen-tolerant hydrogenase
The function of standard hydrogenase is lost when an oxygen atom binds to Ni and Fe atoms. However, the function of some types of hydrogenase is not lost; these types are oxygen-tolerant.
<<Figures>>
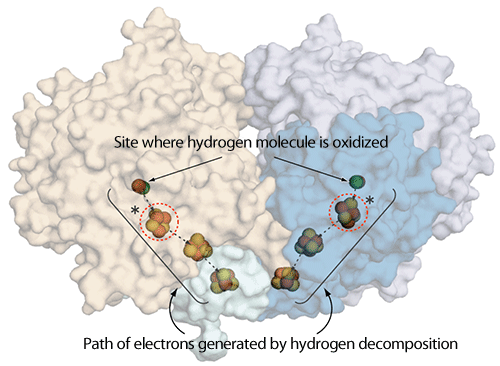
As a result of crystallography, it was found that the protein consists of two hydrogenase molecules (the left molecule is shown in yellow and the right molecule is shown in purple, blue, and light blue; each molecule has one site where hydrogen molecules are oxidized and three Fe-S clusters). Hydrogen molecules are oxidized inside the protein and transferred to a region outside of the protein via a specific path. Sites with a characteristic structural difference depending on the presence of oxygen (Fig. 2) are enclosed by red dotted lines marked with an asterisk.
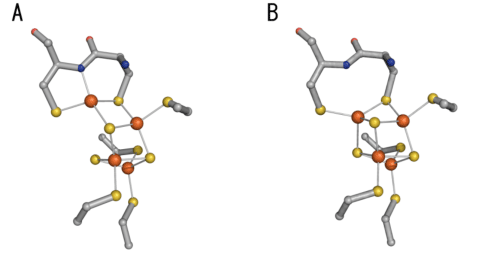
Orange, Fe atom; yellow, S atom; blue, N atom; red, O atom; gray, C atom. Structures A and B are observed in the presence of oxygen; however, only structure B is observed in the absence of oxygen but in the presence of hydrogen.
|
For more information, please contact: |
- Previous Article
- Mechanism Underlying Removal of “Repressive Mark” on Genes by Ubiquitously Transcribed Tetratricopeptide Repeat, X Chromosome (UTX)(Press Release )
- Current article
- X-ray Crystallography of Oxygen-Tolerant Membrane-Bound [NiFe] Hydrogenase (Press Release)