15-fold Upgrade of Fuel Cell Electrode’s Catalytic Activity (Press Release)
- Release Date
- 23 Apr, 2012
- BL15XU (WEBRAM)
National Institute for Materials Science
|
Research teams at National Institute for Materials Science (President: Sukekatsu Ushioda) have successfully achieved a 15-fold upgrade in catalytic activity of fuel cell*1 electrode materials, whereby a newly developed solubilization method for metallic nanoparticles was utilized. The research teams were led by the following researchers: Dr. Hideki Abe (principal researcher, Environmental Remediation Materials Unit), Dr. Hideki Yoshikawa (principal researcher, Nano Characterization Unit/Synchrotron X-ray Station at Spring-8), and Dr. Toru Hara (principal researcher, Surface Physics and Structure Unit). The substantial upgrade of electrode catalytic activity has paved the way for the production of fuel cell materials that require a smaller amount of rare metals. The research results were published on the online publication of Chemical Communication (a journal published by the Royal Society of Chemistry) on March 9. Publication: |
<<Figures>>
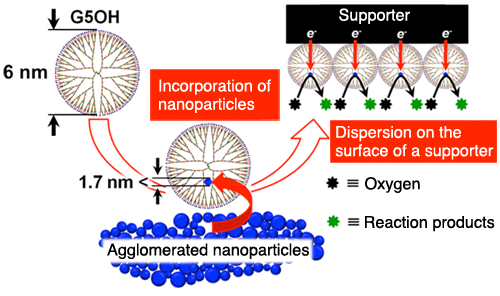
Incorporation of metallic nanoparticles into dendrimers, dispersion on carrier surface, and the catalytic reactions taking place in the fuel cell
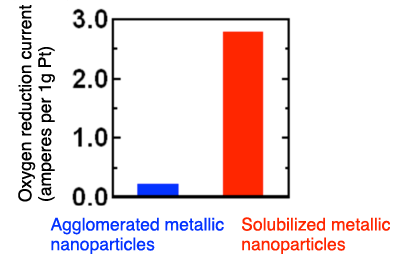
Comparison of fuel cell catalyst activities: agglomerated metallic nanoparticles vs. solubilized metallic nanoparticles
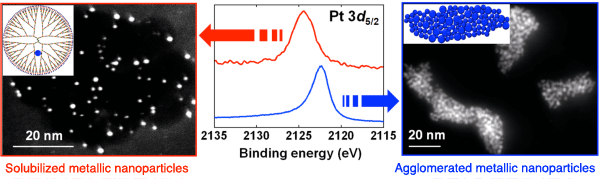
Transmission electron microscope image of solubilized metallic nanopartilces (left) and agglomerated metallic nanoparticles (right). The graphs in the center show the results of the HX-PES measurement for each of the nanoparticles.
<<Glossary>>
*1 Fuel cell
A cell device in which small molecules, such as hydrogen and methanol, are combusted electrochemically with the effect of converting the charge migration within it into an electric current available for use outside the reaction system. The fuel cell is a new technology gathering attention as one of the major next generation energy sources.
|
For more information, please contact:
Dr. Hideki Yoshikawa (National Institute for Materials Science)
Dr. Toru Hara (National Institute for Materials Science) |
- Previous Article
- Visualization of the relation between thermal conductivity and the “rattling” of a guest atom trapped in a cage structure (Press Release)
- Current article
- 15-fold Upgrade of Fuel Cell Electrode’s Catalytic Activity (Press Release)