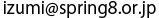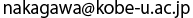Detection of Structural Differences around Specific Atoms in Biomolecules (Press Release)
- Release Date
- 22 Feb, 2013
- BL25SU (Soft X-ray Spectroscopy of Solid)
Japan Synchrotron Radiation Research Institute
Kobe University
Japan Atomic Energy Agency
|
The research groups at the Japan Synchrotron Radiation Research Institute (JASRI), Kobe University, and Japan Atomic Energy Agency (JAEA), in collaboration with the National Institute of Advanced Industrial Science and Technology, succeeded to detect local structural differences in the vicinity of oxygen atoms in several biomolecular amino acids(*1) using soft X-ray(*2) natural circular dichroism(*3) spectroscopy. Soft X-ray absorption spectroscopy has been commonly used to analyze chemical bonding states. In contrast, although soft X-ray natural circular dichroism has been expected to obtain the structural information of biomolecules, research on it has made slow progress due mainly to the extreme difficulty of measurement. In this research, high-speed switching between right and left circularly polarized(*4) soft X-rays - generated by the twin helical undulators(*5) installed in the beamline BL25SU at SPring-8(*6) - was fully exploited to reduce the noise level down to 0.1% or less of absorption intensity, leading to drastically enhanced accuracy of the soft X-ray natural circular dichroic spectrum. The measurement technique enables high sensitivity detection of small differences in types of steric conformations that have been hitherto inaccessible by conventional absorption spectroscopy. Subtle differences in a variety of amino acid side chains(*7) were detected by using soft X-ray natural circular dichroism spectroscopy. The measurement technique places expectations on developing a range of applications in a variety of fields, e.g. detailed structural elucidation of proteins and biomolecules, and applications in drug discovery. The research results were published in the Journal of Chemical Physics, a journal published by the American Institute of Physics, on February 21, 2012 (EST). Publication: |
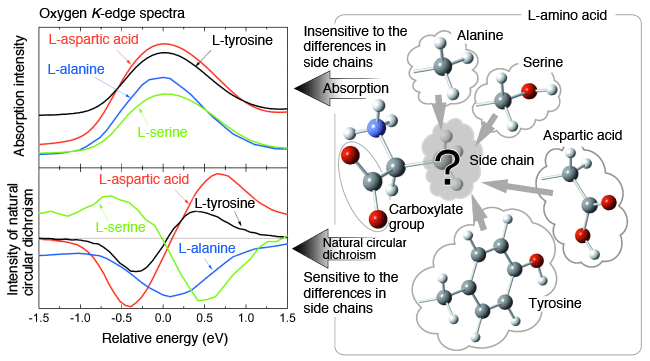
<<Figures>>
(Right) The structure of L amino acid. Its side chain varies depending on the type of the amino acid. Absorption spectra (upper left), and natural circular dichroism spectra (lower left) of the carboxylate oxygen (common part in all amino acids). The horizontal axis represents relative energy in reference to the value at the peak of the absorption spectra (defined as 0eV). Actual energy at the peak is equivalent to approximately 532eV.
<<Glossary>>
*1 Amino acids
Biomolecules having an amino group (-NH2) and a carboxyl group (-COOH). Amino acids in solid state are known to exhibit a zwitterion state, in which both amino and carboxyl groups are ionized (-NH3+ and -COO-). Some amino acids occur two enantiomers, L- and D-type, which are mirror image of each other. In living organisms, L-type is dominant. Proteins consist mainly of 20 amino acids.
*2 Soft X-ray
An X-ray with such comparatively low energy that it hardly penetrates through air. The energy range is suited for analyzing the carbon, nitrogen, and oxygen atoms, which constitute biomolecules, selectively.biological molecules individually, i.e. carbon, nitrogen, and oxygen.
*3 Natural circular dichroism
Any molecule (or any material in general) absorbs light in a specific energy range. Natural circular dichroism refers to the differential absorption of left and right circularly polarized light of which energy are same. Natural circular dichroism measurement, especially in the UV range, is widely used to determine the structure of proteins and other molecules.
*4 Circularly polarization
Light is a transverse electromagnetic wave with mutually perpendicularly oscillating electric and magnetic fields. Light is said to be circularly polarized if its direction of field oscillation - electric and magnetic - rotates in a plane perpendicular to the direction of propagation. It is right-circularly polarized if its electric field rotates clockwise, seen from the destination of propagation, and left-circularly polarized otherwise.
*5 Twin helical undulators
“Twin helical undulators” consists of two helical undulators which provided right and left circularly polarized soft X-rays and it was used as a light sourse in this research. The system allows high-speed switching two polarization states.
*6 SPring-8
A RIKEN facility located in Harima Science Garden City (Hyogo prefecture) is capable of producing the world's highest intensity synchronous radiation. The management and promotion of utilization of this facility are undertaken by JASRI. The name “SPring-8” comes from “Super Photon ring-8GeV.” An electron flying at nearly the speed of light, if deflected from its original trajectory through the effect exerted by a magnet, emits an electromagnetic wave in a direction tangential to its trajectory, which is called radiation light (or synchrotron radiation). At present, there are three “3rd Generation” large scale synchronous radiation facilities in the world: SPring-8 (Japan), APS (USA) and ESRF (France). The acceleration energy available at SPring-8 (8 billion electron volts) enables the provision of an extremely wide spectrum of radiation light: from far infrared to visible, vacuum ultraviolet, and soft X-ray up to hard X-ray. SPring-8 provides a theater for collaborative works involving researchers inside and outside Japan, and the research conducted at this facility cover such diverse areas as material science, geoscience, life science, environmental science, and various applications in industrial sectors.
*7 Side chain
The carbon atom at the center of an amino acid in living organisms has four chemical bonds connected to four atoms or functional groups. Three of them are invariably a hydrogen atom, a carboxyl group, and an amino group. But the last one varies from each amino acid and it is generally called a side chain.
|
For more information, please contact: Prof. Kazumichi Nakagawa (Kobe University) Dr. Akane Agui (JAEA) |
- Current article
- Detection of Structural Differences around Specific Atoms in Biomolecules (Press Release)