New Material Capable of Trapping and Decomposing Specific Gases - Remarkable potential of pores
What are porous materials?
Porous materials are frequently used in products around us. Porous materials, as the name indicates, have many minute pores. Typical examples include activated carbon and zeolites*1 (Fig. 1). For example, activated carbon is frequently used to remove odors from refrigerators and automobiles because the minute pores on the surface of activated carbon adsorb gas molecules, a source of odors. In addition, porous materials are widely used as separators during petroleum refinery and as water purification materials.
The size and properties of pores vary among porous materials, and their applications depend on these factors. If we can control the size and properties of these pores, the range of applicability of porous materials will expand tremendously. For example, they can be used to improve the global environment by removing pollutants from the environment or to generate resources by extracting particular molecules from air.
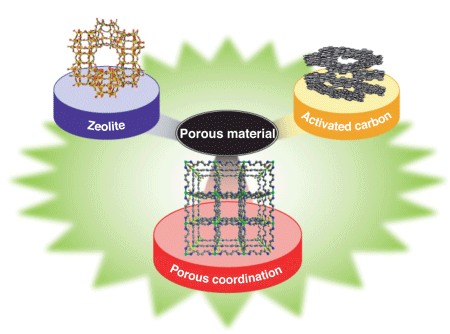
Fig. 1
The pore size of activated carbon is nonuniform, which is advantageous for purification and removing odors from water. In contrast, the pore size of zeolites is uniform and the screening of specific molecules is possible but the range of pore size is limited because zeolites are inorganic. Porous coordination polymers have functions that are completely different from those of conventional porous materials, whose application and usage have been limited. Activated carbon and zeolites have been used for approximately 3,600 and 250 years, respectively, whereas research on porous coordination polymers started only 14-15 years ago, and their range of applications is expected to increase in the future.
Developing a skeleton using flexible organic materials
Susumu Kitagawa, a professor of Kyoto University, has created a path toward realizing the above vision. In 1992, shortly after becoming a professor of Tokyo Metropolitan University, he was involved in research on the synthesis of coordination polymers, in which metal ions and organic molecules are interchangeably coordinated. One day, a chance to further develop his research arose. At a conference, he presented the results of his research on a new coordination polymer that he had synthesized. A researcher from Osaka Gas Co., Ltd. commented that there must be small pores between the coordinated molecules. He also added “if gas molecules are adsorbed in the pores, the coordination polymer can be used as a new porous material,” then asked if he could participate in the research, which was the beginning of this study.
“The coordination polymer presented at the conference could not be used as it was, because it collapsed when the solvent, or a liquid used for synthesis, was removed from the pores. I was so excited when a coordination polymer that didn't collapse upon the removal of the solvent was obtained after about two years of trial and error,” recounts Professor Kitagawa. “At first, many researchers doubted this finding because they believed that organic materials could not be used as a skeleton for porous materials because of their softness. We were able to demonstrate that organic materials could be used in this way after we published a paper in 1997.”
The material that Professor Kitagawa first synthesized was called a porous coordination polymer (Fig. 2). Today, these polymers are a highly competitive area of research in which more than 2,000 papers are published per year worldwide.

Fig. 2 Crystal powder of porous coordination polymer (left) and schematic image (right)
Ten billion nanopores exist in a cube of side 1 μm (a micrometer is one-millionth of a meter). Gas molecules (red spheres) are adsorbed into the nanopores.
Advantages of porous coordination polymers
One of the advantages of porous coordination polymers is their ease of synthesis, which can be understood by considering the Lego-type blocks that you might have played with in childhood. It takes a long time to make an object by building it block by block. However, a porous coordination polymer with an orderly structure is automatically built in a few minutes simply by mixing solutions containing metal ions and organic molecules. In addition, no thermal energy or application of pressure is required. We can also design porous coordination polymers with various structures because information on the binding sites of metal ions can be applied to the organic molecules.
The structure of porous coordination polymers is flexible, unlike that of activated carbon and zeolites, which are inorganic and inflexible and whose structures hardly change. However, the structure of the porous coordination polymers dynamically changes. For example, a certain porous coordination polymer that can detect the approach of a specific gas molecule opens its pores when such a molecule approaches. “The flexibility of porous coordination polymers is a novel property. In conventional materials, molecules are separated using differences in their size and boiling point. However, there are many molecules that cannot be separated in this way. The materials we are synthesizing have high selectivity as if we pick something with fingers, and I believe that we can realize various separation processes for molecules,” says Professor Kitagawa.
For the design of such functional porous coordination polymers, the SPring-8 beamlines are indispensable. Strong synchrotron radiation and an observation system that enables simultaneous measurement are necessary to observe the dynamic structural changes of small crystals as small as 1 μm. “The images obtained by ordinary X-ray diffractometers are blurred; however, we can clearly observe the changing state of crystals by a method similar to taking flash photographs using SPring-8 beamlines. The prediction of which structures can adsorb which molecules is not yet satisfactory. The information obtained through structural analysis using the SPring-8 beamlines is indispensable for the design of new materials,” says Professor Kitagawa.
Control of adsorption by light irradiation
In July 2010, Professor Kitagawa developed a porous coordination polymer in which the adsorption of gas molecules is controlled by light irradiation. Without the irradiation of ultraviolet light, various gas molecules can freely enter and exit the nanopores of the porous coordination polymer. Upon the irradiation of ultraviolet light, however, only oxygen and carbon monoxide molecules can be selectively adsorbed and decomposed. This is the first porous material with such a property. What mechanism underlies this property?
The key to this mechanism is nitrenes. A nitrene is a nitrogen atom that does not satisfy the octet rule;*2 it tries to become stable by depriving electrons of oxygen and carbon monoxide molecules. If we can incorporate the nitrenes into the nanopores of a porous coordination polymer, the selective adsorption of only oxygen and carbon monoxide molecules will become possible. However, it is difficult to incorporate highly reactive nitrenes into the nanopores of a porous coordination polymer.
To solve this problem, Professor Kitagawa and colleagues covered a nitrene with a nitrogen molecule (as a lid) to confine the reactivity of the nitrene and form a molecule called azide (-N3). Using azide, a new porous coordination polymer was synthesized (Fig. 3). The nitrogen molecule used as a lid is easily removed by the irradiation of ultraviolet light. Namely, by irradiating ultraviolet light, the lid can be removed on demand to adsorb oxygen and carbon monoxide molecules, by which carbon monoxide is decomposed into nontoxic materials. Also, nitrenes are difficult to observe because of their high reactivity and instability. Thanks to the SPring-8 X-rays, the research group succeeded in directly observing the ordered arrangement of nitrenes on the surface of the nanopores of the porous coordination polymer.
“I think that our porous coordination polymer can be applied to the removal of toxic carbon monoxide. The polymer we created can be used with various chemical species other than nitrenes. I believe that there will be a major ripple effect as a result of this breakthrough,” says Professor Kitagawa. He also added, “To cope with energy problems, we will have to utilize materials around us rather than special resources. What we are aiming at is ‘the hermetic art of gases,’ namely, the generation of resources by separating, storing, and converting specific molecules in air using porous coordination polymers. We are only at the starting point of realizing this. Our challenge for the future is how to relate our technique with new innovations.” Increasing expectations are likely to be placed on Professor Kitagawa and colleagues, who contribute to solving problems related to both energy and resources through their research.
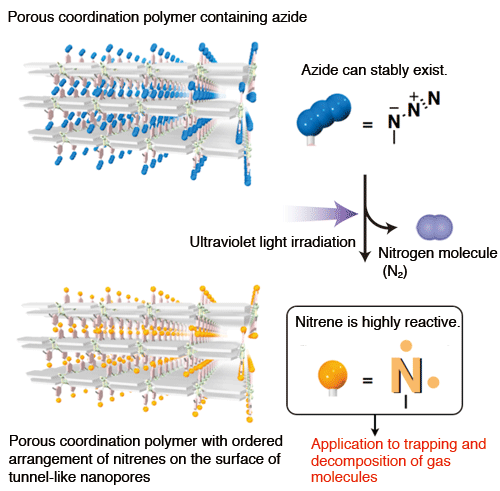
Fig. 3
Nitrogen molecules are removed and nitrenes appear when ultraviolet light is irradiated on the porous coordination polymer synthesized using azides. As a result, oxygen and carbon monoxide molecules are selectively adsorbed and decomposed.
Column: While waiting for data analysis
 In 1989, before Professor Kitagawa started his research on porous coordination polymers, there were no giant beamlines such as SPring-8 or computer programs to analyze related data, which made X-ray structural analysis very laborious work. “I was used to go to the computer center of Kyoto University to input X-ray diffraction data on developed coordination polymers into the computer. However, I had to wait for two to three hours before I could get the calculation results because it took a long time for the calculation. I had nothing to do during this time so I chatted with students. One of the students said ‘There are pores in a honeycomb structure,’ while observing the progress of one of the calculations. This was the first time that I noticed the existence of pores in the coordination polymer. But I didn't think of utilizing the pores then,” recalls Professor Kitagawa. It is impressive that an inspiration obtained during a conversation to kill time has grown to research theme attracting global attention.
In 1989, before Professor Kitagawa started his research on porous coordination polymers, there were no giant beamlines such as SPring-8 or computer programs to analyze related data, which made X-ray structural analysis very laborious work. “I was used to go to the computer center of Kyoto University to input X-ray diffraction data on developed coordination polymers into the computer. However, I had to wait for two to three hours before I could get the calculation results because it took a long time for the calculation. I had nothing to do during this time so I chatted with students. One of the students said ‘There are pores in a honeycomb structure,’ while observing the progress of one of the calculations. This was the first time that I noticed the existence of pores in the coordination polymer. But I didn't think of utilizing the pores then,” recalls Professor Kitagawa. It is impressive that an inspiration obtained during a conversation to kill time has grown to research theme attracting global attention.
Glossary
*1 Zeolites
Zeolites are the general term for aluminosilicates, which have many pores in the crystals. Zeolites are naturally formed over approximately seven millions of years; they can also be formed artificially. Zeolites are used in various fields, such as deodorants, ion exchange, catalysts, and adsorbents.
*2 Octet rule
The octet rule is a rule of thumb that a chemically bonded atom is stable when the number of its outermost electrons is eight. Nitrenes have only six outermost electrons and try to deprive electrons of other atoms and molecules. Therefore, they are highly chemically reactive and change into other molecules by reacting with oxygen and carbon monoxide molecules.
Interview and original text by Chisato Hata (Sci-Tech Communications Incorporated)
This article was written following an interview with Professor Susumu Kitagawa, Deputy Director of the Institute for Integrated Cell-Material Sciences (iCeMS), Kyoto University.