Ordering Is Required for Glass-Forming Liquids -Complete clarification of atomic and electronic structures of extremely ‘fragile’ liquid at 2800 ℃- (Press Release)
- Release Date
- 18 Dec, 2014
- BL04B2 (High Energy X-ray Diffraction)
- BL08W (High Energy Inelastic Scattering)
Japan Synchrotron Radiation Research Institute (JASRI)
Japan Advanced Institute of Science and Technology (JAIST)
Japan Aerospace Exploration Agency (JAXA)
Gakushuin University
The University of Tokyo
Yamagata University
|
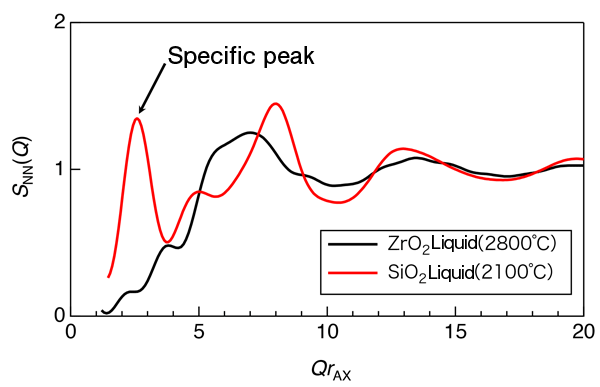
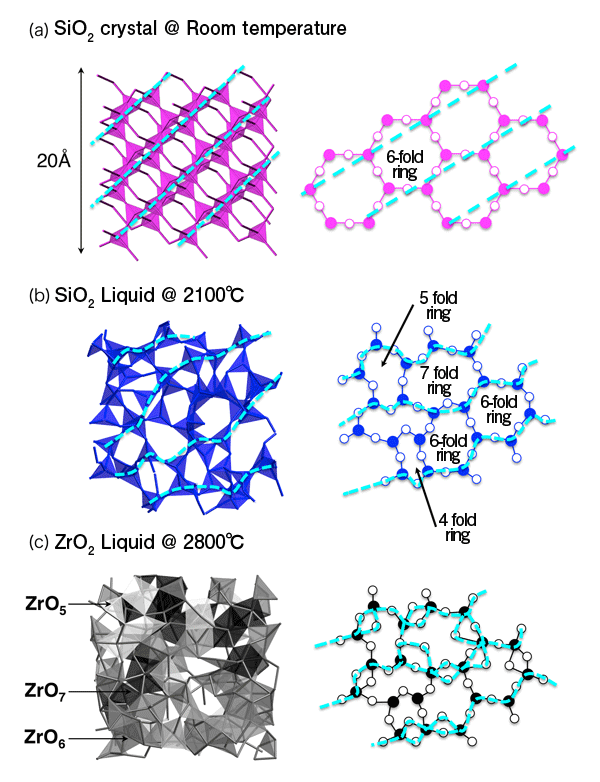
An international joint research team examined the atomic and electronic structures of a non-glass-forming liquid and clarified, for the first time in the world, that the liquid is extremely ‘fragile’ because of its highly disordered atomic and electronic structures and hence cannot form a glass. The team consisted of researchers from JASRI, JAIST, Tampere University of Technology in Finland, JAXA, Gakushuin University, the University of Tokyo, and Yamagata University. These achievements were obtained through experiments using high-brilliance high-energy synchrotron radiation X-rays at SPring-8*1 and large-scale simulation using supercomputers at the IT Center for Science Ltd., in Finland, Forschungszentrum Jülich in Germany, and at JAIST. Publication: |
<<Figures>>
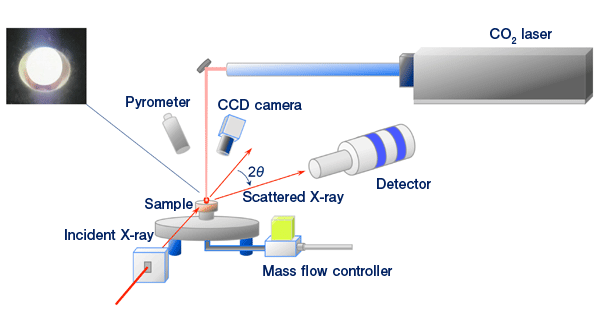
A sample is heated and melted by a CO2 gas laser, levitated by a gas ejected from a conical nozzle, and irradiated with high-energy synchrotron radiation X-rays in the diffraction experiment. The upper left figure shows a photograph of the levitated high-temperature oxide melt.
<<Glossary>>
※1 SPring-8
SPring-8 (Harima Science Park, Hyogo, Japan) is a synchrotron radiation facility that provides the world's highest brilliance radiation. It’s owned by Riken and run by JASRI. The name "SPring-8" is from "Super Photon ring-8 GeV". Synchrotron radiation is a electromagnetic wave radiated when charged particles are forced to bend in magnetic fields. Synchrotron radiation from SPring-8 is widely used for the studies of nano-technology, bio-technology and industrial purposes.
※2 Melt
A melt is a synonym for a liquid. In particular, metallic and oxide liquids with high melting points are referred to as melts.
※3 Containerless gas levitation method (see Fig. 1)
An inert gas (e.g., Ar, N2) is blown from a conical nozzle to a sample upward in the vertical direction to levitate the sample without using a container. The sample is irradiated with a CO2 laser that can easily achieve a high temperature exceeding 2000°C. In this method, there is no risk of the melt being contaminated with the dissolved materials of the container and the liquid state is maintained (supercooled liquid state) at temperatures even below the melting point because of the absence of a heterogeneous interface between the melt and the container (crystal). The absence of the interface also prevents the formation of nuclei for the crystallization of the liquid, enabling the glass formation of materials with a low glass-forming ability.
※4 High-energy X-ray diffraction
When a material with an ordered atomic configuration is irradiated with X-rays as electromagnetic waves, the waves scattered at each atom interfere with each other, causing a strong diffraction wave (diffracted X-ray) to propagate in a specific direction. This phenomenon is called X-ray diffraction and can be used to analyze the atomic arrangement in materials. SPring-8 can generate high-energy X-rays with a high transmittance, which enables high-energy X-ray diffraction.
※5 Fragile liquid
A “fragile liquid” is a liquid that is difficult to form a glass and its opposite is a “strong liquid”. These terms were introduced in Science in 1995 by Austen Angell, who examined the temperature dependence of the viscosity of liquids from high temperature to the glass transition temperature. He defined liquids whose viscosity rapidly changes with decreasing temperature during solidification as fragile liquids and liquids whose viscosity moderately changes (or is originally high) as strong liquids. Since then, these concepts have been widely used as indices of the glass forming ability of liquids.
|
For more information, please contact: |
- Current article
- Ordering Is Required for Glass-Forming Liquids -Complete clarification of atomic and electronic structures of extremely ‘fragile’ liquid at 2800 ℃- (Press Release)