Structure determination of oxygen-evolving photosystem II from T. vulcanus at 3.7 angstrom resolution
Inquiry number
SOL-0000001163
Beamline
BL41XU (Macromolecular Crystallography I)
Scientific keywords
| A. Sample category | biology, medicine |
|---|---|
| B. Sample category (detail) | biomolecule, crystal, protein |
| C. Technique | X-ray diffraction |
| D. Technique (detail) | single crystal |
| E. Particular condition | low-T (~ liquid N2) |
| F. Photon energy | X-ray (4-40 keV) |
| G. Target information | chemical state, molecular structure, structure analysis, function and structure, charge density |
Industrial keywords
| level 1---Application area | environment, Pharmaceuticals |
|---|---|
| level 2---Target | catalysis, drug design, process analytical technology (PAT) |
| level 3---Target (detail) | protein, drug |
| level 4---Obtainable information | interatomic distance, supra-molecular assemblies |
| level 5---Technique | diffraction |
Classification
A80.32 organic material, A80.50 Pharmaceuticals, M10.10 single crystal diffraction
Body text
In the initial step of photosynthesis, water molecule is splitting to an oxygen molecule and electrons needed to later steps. Photosystem II is a membrane protein complex to do this reaction.
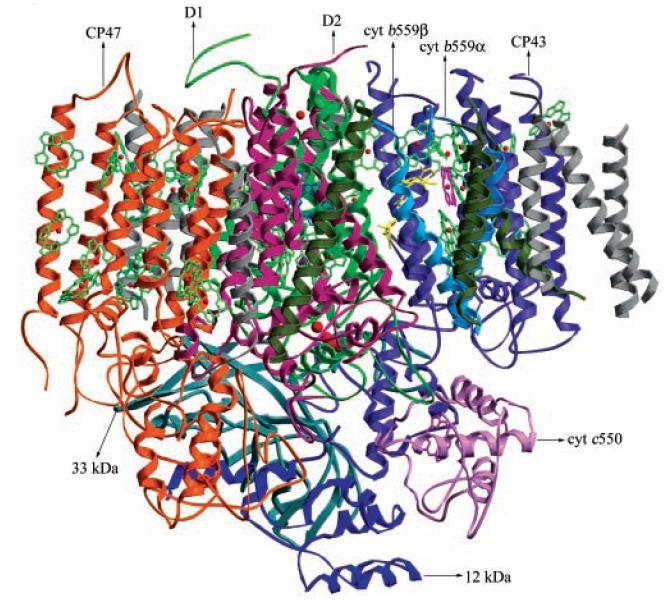
In this case, crystal structure of oxygen-evolving photosystem II from thermophilic cyanobacteria T. vulcanus was determined at 3.7 Å resolution (Fig. 1).
Generally, most crystals of membrane protein have only weak ability to diffract X-ray. The unit cell of this crystal is large, 129.7x226.5x307.8 Å. High brilliant X-ray from undulator of BL41XU was indispensable to collect the good X-ray diffraction data.
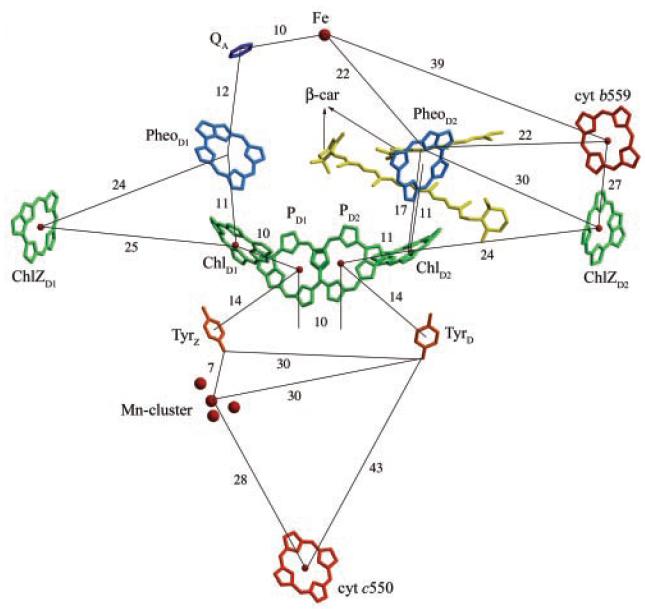
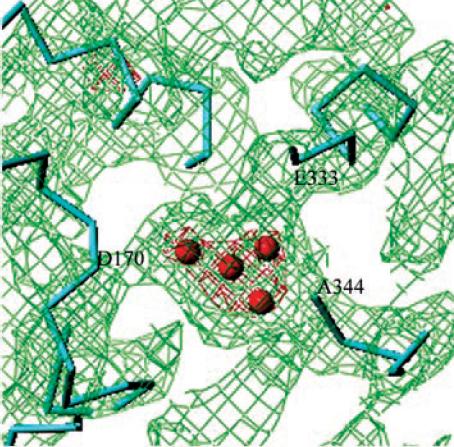
Three-dimensional pathway of light-induced electron transfer (Fig. 2) and constitution of water-splitting Mn cluster (Fig. 3) were revealed.
Figure 1. Structure model of oxygen-evolving photosystem II from T. vulcanus
Figure 2. Arrangements of chlorophylls and other cofactors and their relative distances
Figure 3. Mn cluster and the electron density surrounding it
[ N. Kamiya and J.-R. Shen, Proceedings of National Academy of Science of the USA 100, 98-103 (2003), Fig. 1, 3, 4,
©2003 National Academy of Science ]
Source of the figure
Original paper/Journal article
Journal title
N. Kamiya, J. R. Shen Proc. Natl. Acad. Sci. U. S. A. 100, 98-103 (2003)
Figure No.
1,3,4
Technique
Source of the figure
No figure
Required time for experimental setup
hour(s)
Instruments
| Instrument | Purpose | Performance |
|---|---|---|
| Protein Crystal Diffractometer | To record diffraction data |
References
| Document name |
|---|
| N. Kamiya, J. R. Shen Proc. Natl. Acad. Sci. U. S. A. 100, 98-103 (2003) |
Related experimental techniques
Questionnaire
The measurement was possible only in SPring-8. Impossible or very difficult in other facilities.
This solution is an application of a main instrument of the beamline.
Ease of measurement
Middle
Ease of analysis
Middle
How many shifts were needed for taking whole data in the figure?
More than ten shifts