An Atomic-Level Analysis of the Mechanism of Proteins Controlling the Iron Concentration inside a Cell (Press Release)
- Release Date
- 07 Aug, 2012
- BL26B1 (RIKEN Structural Genomics I)
- BL26B2 (RIKEN Structural Genomics II)
- BL41XU (Structural Biology I)
Institute for Molecular Science, National Institutes of Natural Sciences
|
A research group under the direction of prof. Shigetoshi Aono of the Institute for Molecular Science (Okazaki Institute for Integrative Bioscience), National Institutes of Natural Sciences, together with a research group under the direction of chief scientist Yoshitsugu Shiro of the RIKEN SPring-8 Center, RIKEN Harima Institute, through biochemical experiments and X-ray crystal structural analysis, has successfully identified the protein that operate as a switch to regulate the expression of a heme efflux system upon sensing toxic free hemes, and the group has also identified the details of the mechanism whereby the intercellular heme concentration is kept constant. Publication: |
<<Figures>>
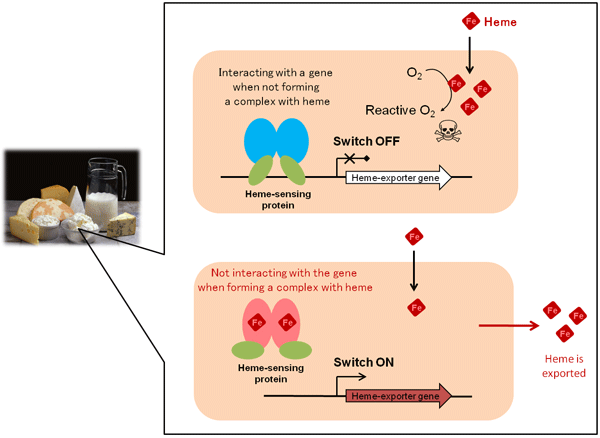
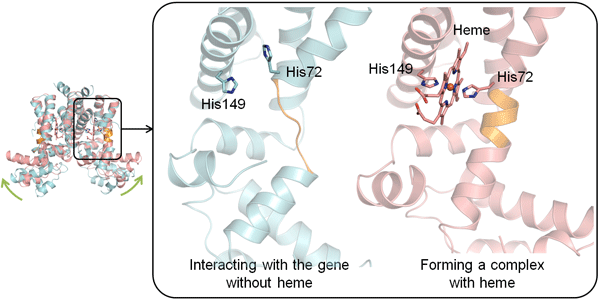
The light pink rectangle represents a Lactococcus lactis cell.
The figure on the left combines the total structure in both cases.
<<Glossary>>
*1 Heme
A heme is a compound consisting of an iron atom at the center of a planar ring-shaped molecule called “porphyrin.” Hemes can be classified into certain types by the type and position of the porphyrin ring configuration. A protein that can function only when it has taken in a heme is called a heme protein (or hemoprotein) and is normally red in color. Well-known examples include hemoglobin, which carries oxygen, cytochromes, which are involved in electron transfer, and peroxidase, which activates enzymes.
*2 Reactive oxygen
A comprehensive term referring to any chemical species in which the oxygen molecules in the atmosphere have become chemically highly reactive. Examples include OH· (hydroxyl radical), O2·- (superoxide anion radical), and H2O2 (hydrogen peroxide). These cause oxidation damage to biological molecules, such as DNAs, lipids, and proteins, causing them to lose their proper physiological functions.
*3 X-ray crystal structural analysis
A method of determining the structure of molecules that constitute a crystal. An X-ray, which is a type of light with a short wavelength, is radiated onto a crystal, in which matter is properly aligned with regularity, and the intensity of the diffracted X-ray is carefully studied. Using this method, the 3-dimensional structure of many biological molecules has been determined.
*4 SPring-8
A RIKEN facility located in Harima Science Garden City (Hyogo prefecture) is capable of producing the world's highest intensity synchronous radiation. The management and promotion of utilization of this facility are undertaken by JASRI. The name “SPring-8” comes from “Super Photon ring-8GeV.” An electron flying at nearly the speed of light, if deflected from its original trajectory through the effect exerted by a magnet, emits an electromagnetic wave in a direction tangential to its trajectory, which is called radiation light (or synchrotron radiation). At present, there are three “3rd Generation” large scale synchronous radiation facilities in the world: SPring-8 (Japan), APS (USA) and ESRF (France). The acceleration energy available at SPring-8 (8 billion electron volts) enables the provision of an extremely wide spectrum of radiation light: from far infrared to visible, vacuum ultraviolet, and soft X-ray up to hard X-ray. SPring-8 provides a theater for collaborative works involving researchers inside and outside Japan, and the research conducted at this facility cover such diverse areas as material science, geoscience, life science, environmental science, and various applications in industrial sectors.
|
For more information, please contact: |
- Previous Article
- Successful Visualization of “Tilted” Spins at the Junction Interface of Dissimilar Magnetic Bodies (Press Release)
- Current article
- An Atomic-Level Analysis of the Mechanism of Proteins Controlling the Iron Concentration inside a Cell (Press Release)